Serialized Transaction Information (STI) is the serialized product data required by DSCSA to support end-to-end drug traceability. It allows trading partners to track, verify, and exchange unit-level product information with confidence.

On Oct. 9, 2024, the U.S. Food and Drug Administration (FDA) announced new deadlines for DSCSA compliance for eligible manufacturers, distributors and pharmacies.
FDA-Granted Exemptions
The FDA granted exemptions until November 2026 from certain DSCSA requirements to dispensers with 25 or fewer pharmacists and technicians.
The DSCSA serialization dates for compliance depend on the eligibility and election of trading partners under the Oct. 9, 2024, FDA Exemption document. Masters used that exemption to fully comply with its DSCSA serialization obligations by Aug. 27, 2025. Now, Masters shares serialized data for DSCSA in-scope products through the same portals from which you receive lot-level data today.
In order to receive any serialized transaction data for DSCSA in-scope products from Masters, you must have a Sold-To and Ship-To GLN on file with Masters. The serialized transaction data is tied directly to your GLN. If you elect to use a third-party DSCSA solution provider as your repository, you must complete the enrollment process by contacting your sales team member.
Limited serialized transaction data for in-scope product became available to all Masters customers in December 2023. You may access your serialized data the same way that you access your lot-level data from your designated Masters ordering portal or, if you have contracted with a third-party provider, through their service.
Per DSCSA requirements, Masters will maintain a record of your serialized transaction data for DSCSA in-scope products for six years.
Limited serialized data for DSCSA in-scope products became available in December 2023. The availability of a customer’s serialized transaction data for such products depends on the manufacturer’s ability to provide full EPCIS data and the capability at the distribution center of Masters’ shipping affiliate, McKesson, to capture and transmit that serialized data.
Since Aug. 27, 2025, only serialized transaction data for DSCSA in-scope product has been gathered and distributed. Masters maintains lot and serial DSCSA data for six years after the transaction date for such data as required by the DSCSA.
Masters works with our supplier partners to obtain DSCSA serialized data for all DSCSA in-scope products. We also implemented DSCSA serial number capture capabilities through our distribution affiliate, McKesson. These DSCSA serial number capture capabilities enable serial DSCSA data to become available in the Masters portal and enables Masters to send serial DSCSA EPCIS files to customers who request this service, provided we have that serial information from our suppliers.
Masters items shipped in case quantities contain an aggregate 2D barcode containing the information for all the in-scope DSCSA items contained in that case. Mixed packages do not contain a 2D barcode with aggregated information. Recent FDA guidance has suggested that customers should confirm DSCSA transaction data was received.
Masters is making DSCSA transaction data available through our customer-facing portal.
Masters’ data repository called Advanced Track and Trace for Pharmaceuticals (ATTP), stores serialization data from Masters and allow searching, downloading and printing upon requests made through the customer-facing portals, provided that the customer has the required GLN(s).
Masters customers may use our portal link (MastersRX.com) to manage their DSCSA transaction data during the six-year DSCSA record retention requirement. Additionally, customers may opt to have the data transmitted daily using an EPCIS file to their in-house or third-party DSCSA repository for storage.
Masters’ affiliate, McKesson, handles Masters’ DSCSA-required transaction data. Therefore, in order to access DSCSA-required transaction data, Masters customers must agree to McKesson’s “Agreement to Maintain DSCSA-Required Transactional Data”. This agreement is necessary for Masters to manage and retain DSCSA data in McKesson’s customer portals. This licensing agreement appears when you attempt to access the DSCSA data in the portal for the first time and then periodically thereafter to reverify the license agreement. The agreement can also be printed. Below is a copy of the agreement.
AGREEMENT TO MAINTAIN DSCSA-REQUIRED TRANSACTIONAL DATA
THIS AGREEMENT TO MAINTAIN DSCSA-REQUIRED TRANSACTIONAL DATA constitutes the written agreement described in Section 582(d)(1)(B) of the Drug Supply Chain Security Act (“DSCSA”), between McKesson Corporation (“McKesson”) and the company or other entity that you represent (“Dispenser”). This agreement is effective as of the later of July 1, 2015 or the date Dispenser executes this agreement (the “Effective Date”). Dispenser is responsible for maintaining a copy of this agreement and entering into this agreement does not relieve Dispenser of its obligations under Section 582(d)(1) of DSCSA.
Please indicate Dispenser’s agreement and understanding of this agreement to maintain DSCSA transactional data by signing on behalf of Dispenser in the space provided below. By signing on behalf of Dispenser, you represent and warrant that (i) you are duly authorized to and have full legal authority to bind Dispenser to this agreement and (ii) you agree, on behalf of Dispenser, to this agreement.
Previously, when Masters purchased DSCSA in-scope product directly from the manufacturer, we provided a Direct Purchase Statement in lieu of providing the lot number in the lot level DSCSA transaction data. Since Aug. 27, 2025, the transaction data from Masters distributors contains the lot number, serial number and expiration date of DSCSA in-scope product.
Data is currently being stored on all ordering portals. Storage platforms will retain the data for six years.
If older data is required, please reach out to Technical Support through the Contact Us section in your ordering portal.
If you are using a third-party solution provider for your transaction data, you should first contact your sales representative or account manager to complete the enrollment process. Have the following details to share with your representative:
The process takes approximately:
No, DSCSA will not remove the attestation portion of the returns process.
No, the serialized product number will not appear on invoices.
All data that is available to Masters will be accessible through the ordering portals. If data is unavailable when attempting to access EPCIS data from a third-party solution provider, customers should contact their respective provider directly for assistance.
If you are unable to retrieve serialized DSCSA data from the portal, please reach out to Technical Support through the Contact Us section in your ordering portal.
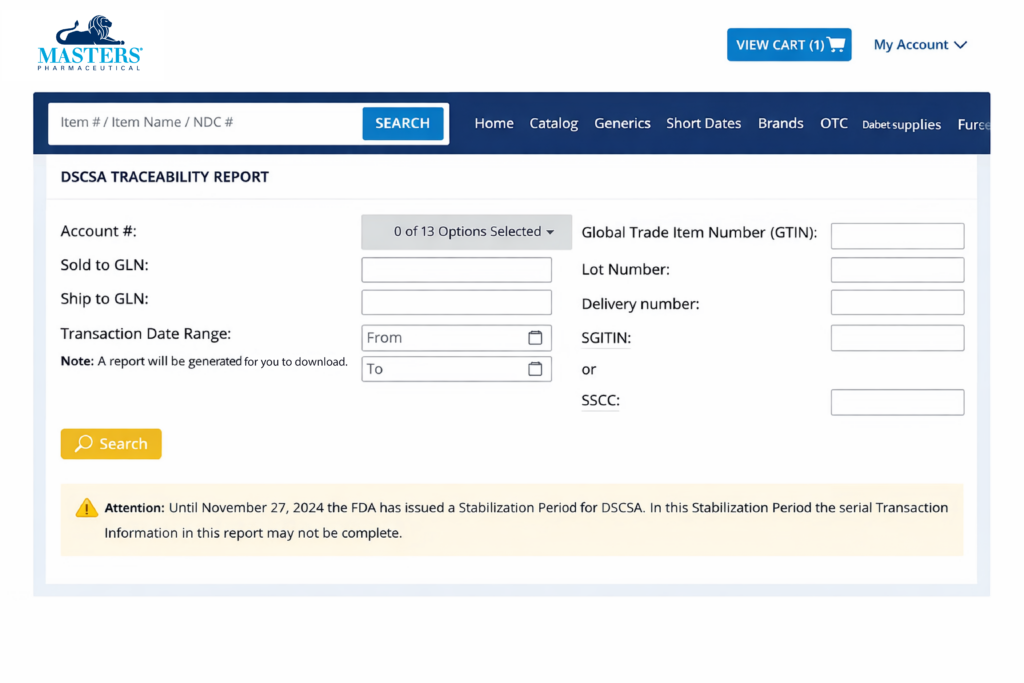
Masters is making DSCSA transaction data available through our customer-facing portals.
On Masters RX.com, you will be able to find your EPCIS data in the DSCSA Serial Traceability Report and DSCSA Lot Traceability Report under My Account as shown below.
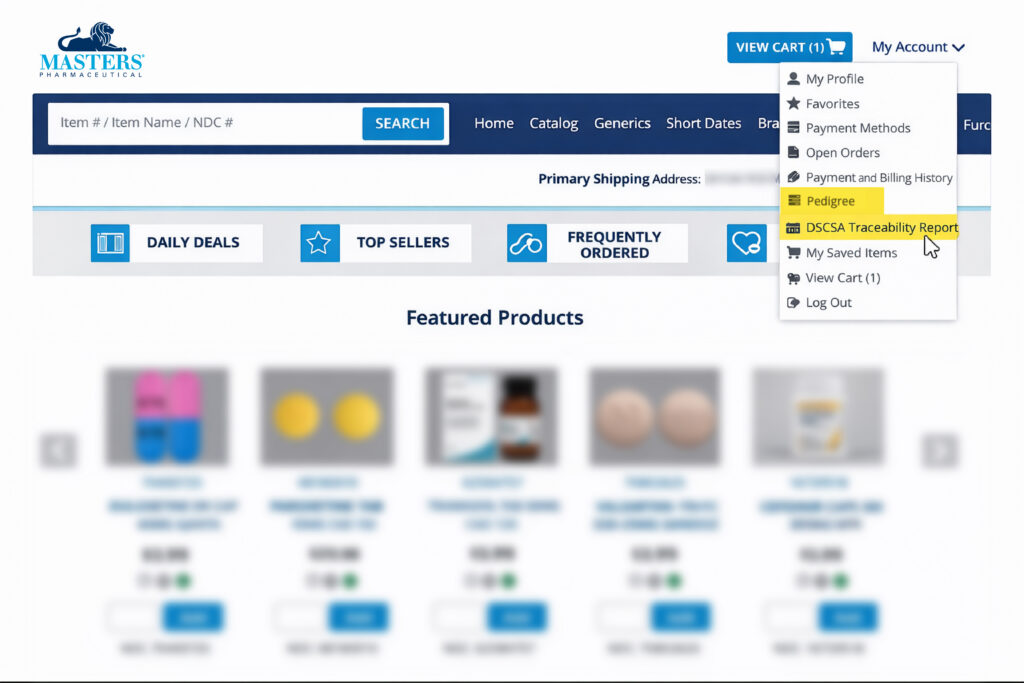
Masters’ data repository, also known as Advanced Track and Trace for Pharmaceuticals (ATTP), stores serialization data from Masters and allow searching, downloading and printing upon requests made through the customer-facing portals, provided that the customer has the required GLN(s). Masters customers may use our portal link to manage their DSCSA transaction data during the six-year DSCSA record retention requirement. Additionally, customers may opt to have the data transmitted daily using an EPCIS file to their in-house or third-party DSCSA repository for storage.
Masters does not have access to the agreement between the customer and the customer’s third-party service provider to manage and store the customer’s DSCSA transaction data. Please contact your service provider directly for a copy of the agreement.
EPCIS data for 340B purchases is provided to the covered entity, not the contract pharmacy. The covered entity can grant access to EPCIS data to their contract pharmacies, but this must be handled by the covered entity and the contract pharmacy.
If there is an error on the EPCIS file it will need to be reported to your Masters representative, and an overage/return or shortage will need to be submitted. Historical EPCIS files cannot be manually altered.
340B contract pharmacies may access DSCSA serialized data only when the covered entity provides explicit authorization for Masters to share it. DSCSA requires that EPCIS data be supplied to the purchasing party, which is typically the covered entity. If a contract pharmacy wishes to receive this data, the covered entity must authorize Masters to share it. This data sharing is optional and not mandated by DSCSA. Masters is currently facilitating outreach to covered entities to support this process and help ensure continuity for contract pharmacies.
The ordering process for drop shipments of DSCSA in-scope products remains the same as before DSCSA requirements were implemented. The manufacturers shipping your product will send you the lot-level DSCSA transaction data. Masters currently does not provide this data. Masters ordering lot-level DSCSA system will indicate a drop shipment occurred but will not have complete DSCSA information as the manufacturer must provide that detail.
To improve the process for serialized DSCSA transaction data, Masters has built functionality to enable it to receive the drop ship serial DSCSA data from the manufacturer and pass it onto the customer. This functionality depends on the manufacturer sending the DSCSA data to McKesson. Even if Masters provides this service, the manufacturer retains the ultimate obligation to send the data.
For drop ship orders of DSCSA in-scope products, Masters owns the product prior to transferring that ownership to the customer. However, since Masters does not possess the product in a drop ship situation, the DSCSA transaction data is created by the shipper. We do expect the shipper to share the data with us which, if received, we will load into our ordering portal or provide to a customer’s third-party DSCSA solution.
Masters has built functionality to enable it to receive the drop ship serial DSCSA data from the manufacturer and pass it onto the customer. This functionality depends on the manufacturer sending the DSCSA data to Masters. Even if Masters provides this service, the manufacturer retains the ultimate obligation to send the data.
We recommend that you refer to the FDA.gov website for more information. The American Society of Health-System Pharmacists (ASHP) and National Community Pharmacists Association (NCPA) also may have relevant resources. For more helpful resources go to DSCSA.pharmacy.
Access to DSCSA data for repackagers requires the purchasing party to grant permission, similar to the process used for 340B covered entities and contract pharmacies. We are currently adapting the 340B agreement process to be applied to repackagers. Once we have the repackager agreement process finalized, your customer will need to complete the agreement authorizing your access to the DSCSA data. We will provide further instructions and support as soon as the process is available.
Explore our Acronym List & Definitions to quickly understand key industry terms—clear, simple, and easy to reference.
